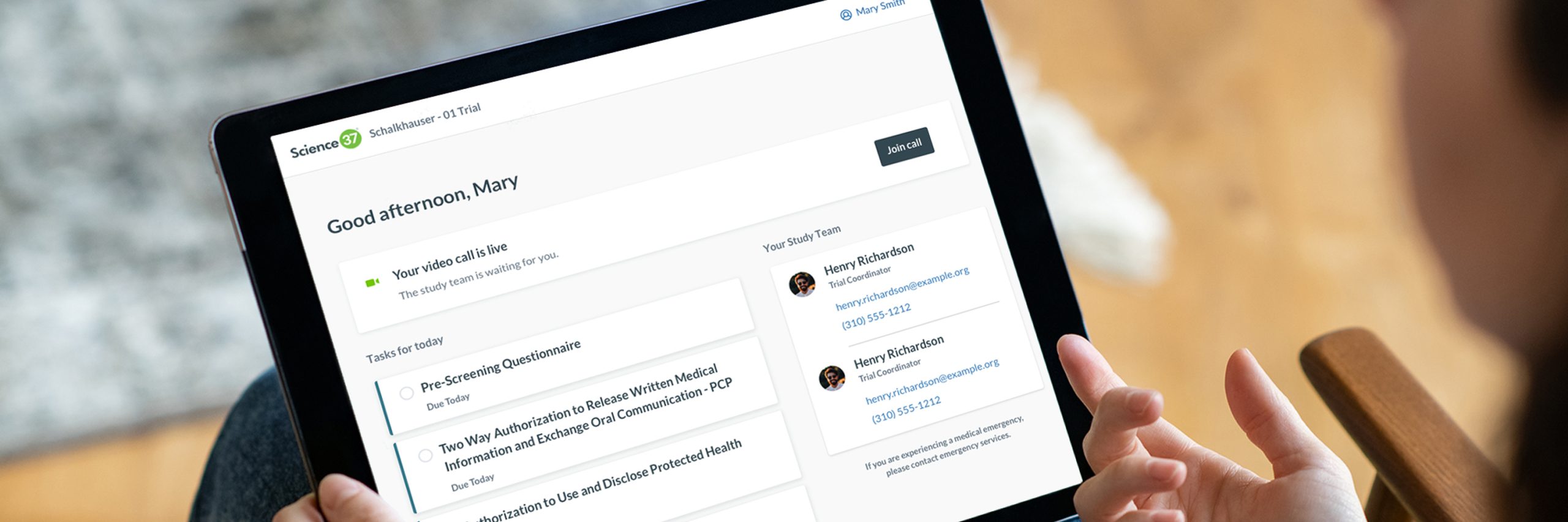
An industry-leading non profit institute was looking to execute a hybrid Phase II Randomized Controlled Study to determine the effectiveness of an interventional therapy.
In this case study we show how Science 37’s unified technology platform and Direct-to-Patient Site (virtual site), helped the sponsor deliver a successful hybrid clinical trial, accelerating timeline. Learn more about the value that this decentralized clinical trial delivered, including: