We’re continuously looking for ways to improve your experience with the Science 37 Platform©. To keep you up to date on key new features and improvements, Product Release Notes will drop into your inbox with every release. Here are highlights of what will come out in this release.
When building forms, data managers can generate automatic queries if a specified field is left blank when a form is submitted. This update will help streamline data management by capturing items that require review without manual intervention.
To enhance patient safety, any adverse event recorded for a patient can trigger an automatic email. Rule-building for this feature will be available in an upcoming release.
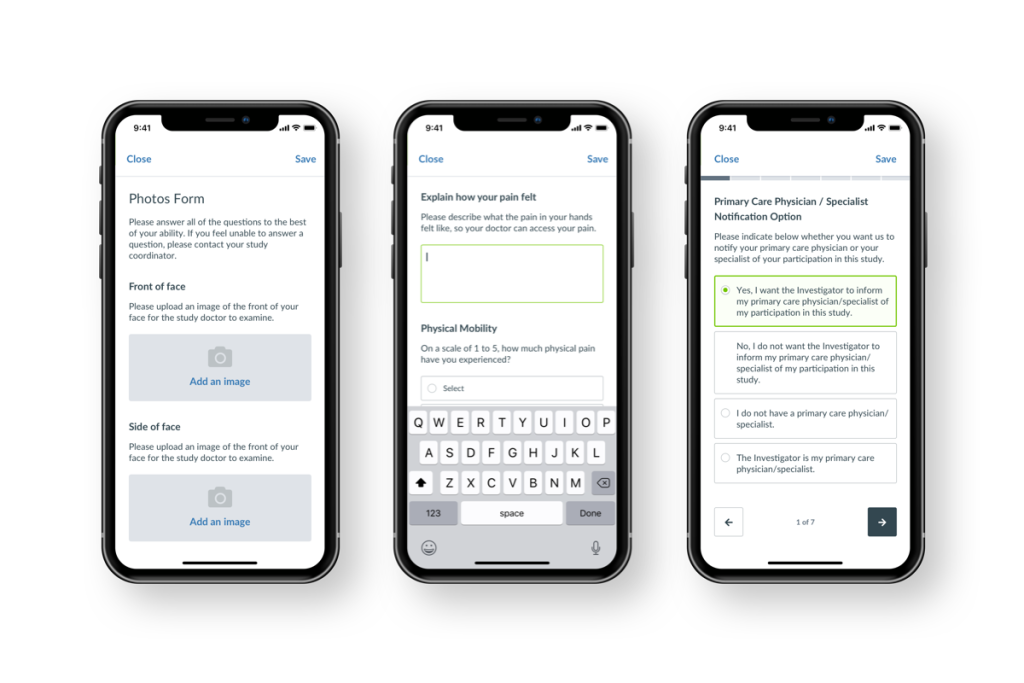
On the Science 37 Clinical Research app, we’ve redesigned the question types—and made a paginated format available—to make completing ePROs (electronic patient-reported outcomes) a smooth, streamlined experience for patients.
v.2.31.0
Target Release Date: June 10, 2020
*Some features may not be available for all users, depending upon study configuration and user roles. This communication (including any attachments) is confidential, may be subject to legal privilege, and is solely for the intended recipient. Any use, duplication, or disclosure of this communication other than by the intended recipient is prohibited. If you have received this communication in error, please notify the sender immediately and delete or destroy this communication and any copies.