Product Release Notes 2.37.0
We’re continuously looking for ways to improve your experience with the Science 37 platform™. To keep you up to date on key new features and improvements, Product Release Notes will drop into your inbox with every release. Here are highlights of what will come out in this release.
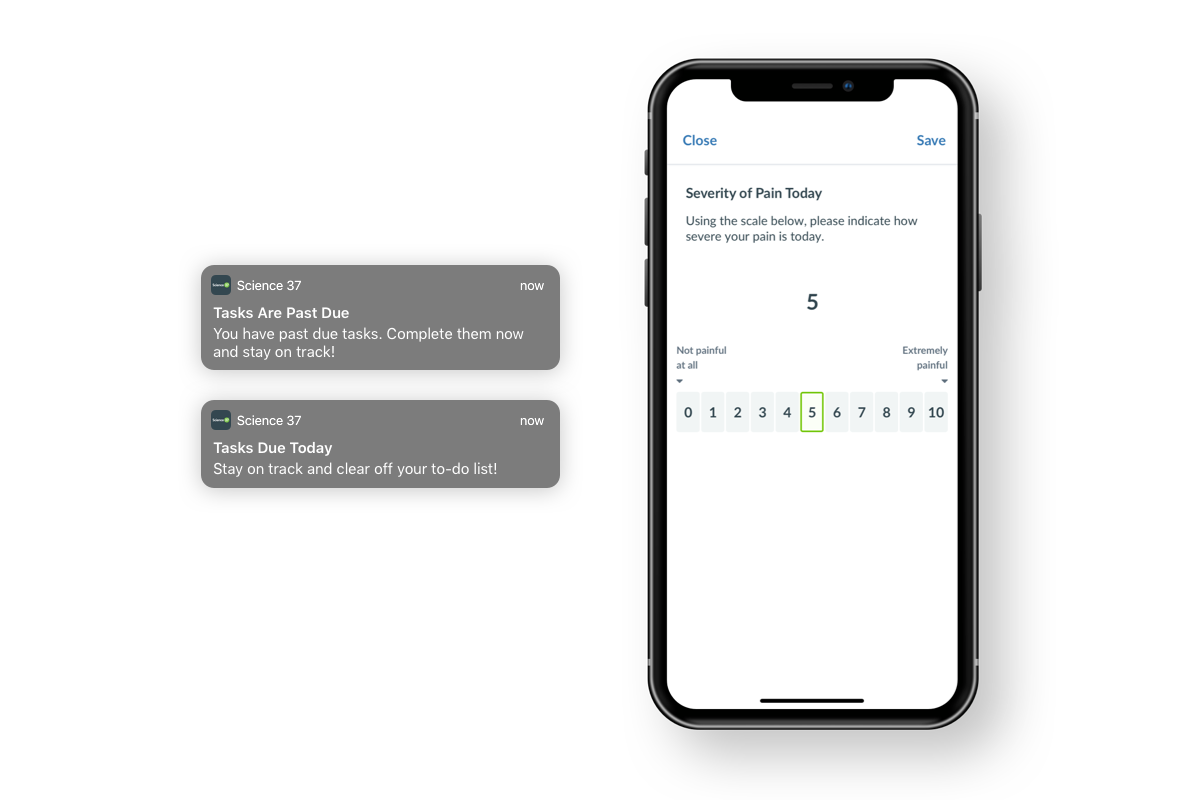
More notifications for patients
We've added Greek language support within our platform and mobile application for patients.
With each release, we're adding new automatic notifications that will help patients stay compliant in their trial, and ease the burden on coordinators to manually remind. With this next release, patients will be notified when:
- they have forms that are due today
- they have forms that are past due
This is only available to the patient in our native apps, available for download on Android and iOS.
More ePRO support: Numeric rating scale
Our new numeric rating scale, or NRS, was designed and built with the patient in mind, while still adhering to the ePRO Consortium Critical Path Institute's guidelines on proper electronic migration of the NRS. With this new question type, we are continuing our expansion of ePRO capabilities that are compliant with the industry's standards, while also making the patient's trial experience as seamless and easy as possible.
Form printing now supports matrices
Coordinators can now print out forms that use matrices, further expanding our form printing capabilities. This feature is available on your forms if printing has been enabled for your trial.
v.2.37.0
Target Release Date: September 14, 2020
*Some features may not be available for all users, depending upon study configuration and user roles. This communication (including any attachments) is confidential, may be subject to legal privilege, and is solely for the intended recipient. Any use, duplication, or disclosure of this communication other than by the intended recipient is prohibited. If you have received this communication in error, please notify the sender immediately and delete or destroy this communication and any copies.